
The materials with the lowest values are all semiconductors with some even reaching down into the near infrared. Most elements have threshold frequencies in the ultraviolet, but a few dip down low enough to be visible, with potassium’s lying somewhere between yellow and green. Each curve has a different intercept on the energy-axis, which shows that threshold frequency is a function of material. Below the threshold frequency photoemission does not occur. The energy-frequency relation is constant for all materials. The two factors affecting maximum kinetic energy of photoelectrons are the frequency of the incident radiation and the material on the surface. Thus, we haveįrom this equation, we see the kinetic energy of an escaped electron depends upon the photons wavelength “λ” only, not on the number of photons. This tells us the product of wavelength and frequency is a constant that is equal to the speed of light. The frequency and wavelength have the relationship: However, this does not happen in experiments, so the wave theory cannot explain the photoelectric effect. The photoelectric effect helps to explain the quantum or particle nature of light and thus supports the argument for the dual nature of light.Īccording to the wave theory, more light shined on a surface would provide more energy to the electrons being ejected from the surface. Light has a dual nature, and hence it has both the properties of a wave and a particle. It is the minimum energy for an electron to leave a metal surface, expressing asįormula for the minimum energy for an electron to leave a metal surface.Īnd this frequency is the metal’s threshold frequency. The ejected electrons maximum kinetic energy can be expressed asįormula for the maximum kinetic energy of an ejected electron. This supports the quantum or particle nature of light. This explained why the energy of photoelectrons was dependent only on the frequency of the incident light and not on its intensity: a low-intensity, high-frequency source could supply a few high energy photons. If you increase the intensity of the light, more photons are colliding with and ejecting electrons, but the energy being carried and transferred by the photons is the same. When the photon has enough energy, its collision with an electron will free it from the surface of a material. Photon Energy Depends on Frequency Not Light Intensity : In the equation, “h” is the Planck constant and “f” is the frequency of incident photon. The amount of energy the light particle carries depends upon its wavelength.įormula for the energy of a light particle. He showed light acts as a quantum particle carrying a discrete amount of energy (E). Using this knowledge Einstein thought light could also be described as discrete photons, not in continuous waves. He realized that the beam of light must be liberating electrons from one metal plate, which are attracted to the other plate by electrostatic forces resulting in a current flow. And it was his contribution to quantum theory - explaining the photoelectric effect - which won Einstein his Nobel Prize in 1921. 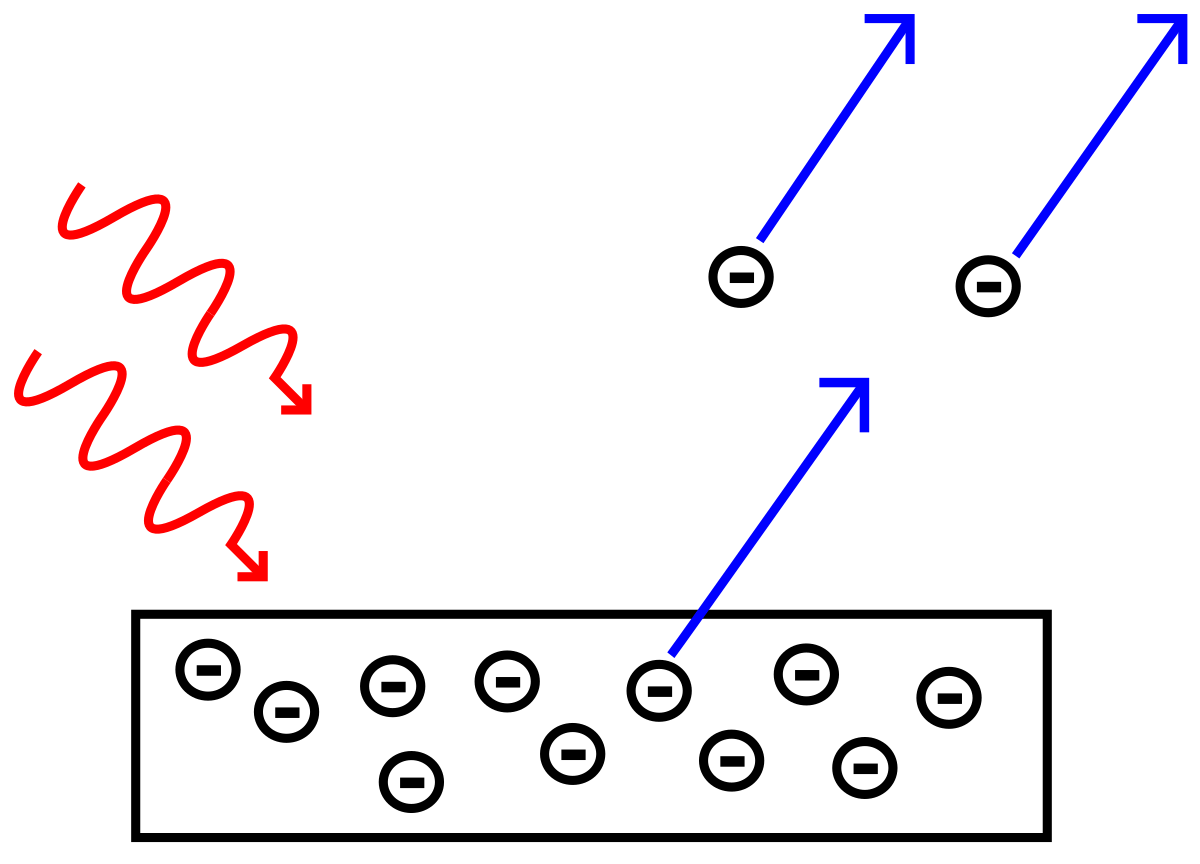
Although Einstein is best known for his theory of relativity, he also played a major role in developing quantum theory. In 20th century physics, two ideas stand out as being totally revolutionary: relativity and quantum theory. The photoelectric effect is the observation that when certain wavelengths of light strike a piece of metal, the electrons in the metal move, creating a current. In 1887, Heinrich Hertz observed a property of light called the photoelectric effect that wave theory could not explain. The lower the energy the light, the less work can be done with it by the atom when it absorbs that light.The Photoelectric Effect - Evidence for Light as a Particle Radio waves cause nuclear spin transitions which is when a proton changes its spin state. Microwaves cause rotational motion where a molecule rotates. 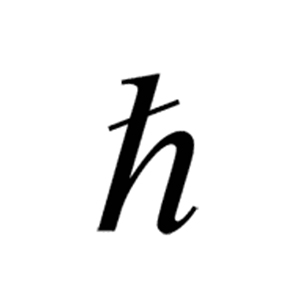
The bonding atoms of a molecule vibrate back and forth like an oscillating spring. Infrared light causes molecular vibrations. The electrons are able to move between the energy levels within the atom, but do not have enough energy to escape. Low energy UV and visible light cause electron transitions. They transfer enough energy to electrons so they can escape from the pull of the atom’s nucleus and turn the atom into an ion. Higher energy light such as gamma rays, X-rays, and high energy UV light cause ionizations. And since energy and frequency are directly proportional, the trend we describe using energy will be the same for frequency. The different effects light has on atoms can best be understood when considering the energies of types of light.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
